What is haematology?
Haematology is the clinical specialty relating to all aspects of blood and its associated structures, specifically lymphoid tissue. In summary, it is concerned with how it forms, its function and pathologies associated with it. As there is no specific organ for haematology, diseases pertaining to haematology will occur either in the bone marrow, lymphoid tissue or intravascular compartment.
Functions of blood
Blood can be regarded as both a tissue and a fluid. It is a tissue because it contains a group of different cells which have specific functions despite being a liquid. Blood and a circulation system to carry that blood is essential for life for anything larger than a primitive multicellular animal.
Broadly speaking, the function of blood is to maintain homeostasis of the internal environment. Therefore, there are many regulatory mechanisms intrinsic to blood. The fact that blood is constantly flowing through the circulatory system means it is primed to adapt to changing conditions. It has many functions which are not replicated by any other organ of the body, including:
- Transportation of oxygen to tissues
- Removal of carbon dioxide, urea and other waste products from tissues
- Delivery of nutrition for cell growth and development: glucose, protein, fats, vitamins, minerals. Certain substance such as glucose and electrolytes are more tightly regulated than others.
- Initiating the immune response
- Thermoregulation by adjusting the rate of heat loss
- Haemostasis and coagulation cascade
Components of blood
Blood has four main components:
- Red blood cells
- White blood cells
- Platelets
- Plasma
Plasma makes up 45-55% of the blood, red blood cells about 45% (higher in men) and white blood cells and platelets a comparably small proportion of 1%.
Red blood cells
Erythrocytes (alternatively referred to as red blood cells) are cells whose primary function is to deliver oxygen to tissues. Erythrocytes are able to do this as they possess haemoglobin, which binds to oxygen to form oxyhaemoglobin. Erythrocytes are specialised for this function as they lose their nucleus and other organelles to make room for as much haemoglobin as possible. Haemoglobin undergoes conformational changes depending on its environment and oxygenated states, which alters how it binds to and releases oxygen. If haemoglobin is deficient or mutated then tissue oxygenation is negatively affected.
It is small and biconcave in shape, giving it a higher surface area to volume ratio that facilitates rapid gas exchange. This shape also allows for increased flexibility, allowing erythrocytes to pass through narrow capillaries. This shape is dependent on membrane proteins. Hence, defects in membrane proteins can change the shape of the red blood cell and affect its functionality.
Erythrocytes survive on average for 120 days in the circulation. After this time, they are consumed by macrophages in the reticuloendothelial system in the spleen or bone marrow. Haemoglobin is metabolised into haem and globin, the former of which is further broken down to bilirubin. Hence in diseases that lead to increased breakdown of red blood cells, bilirubin levels increase overall.
Erythrocytes have surface proteins of which determine someone’s blood type. This is of prime clinical significance for blood transfusions as fatal reactions can occur if the blood types are mismatched.

Source: Keith Chambers, CC BY-SA 3.0, via Wikimedia Commons
White blood cells
White blood cells, also known as leukocytes, have a main function of fighting infection. They generally remain in the circulation but may be upregulated in infective and inflammatory states, or abnormally high due to malignancy. On the other hand, certain bone marrow disease states can lead to lower levels than normal. There are two broad categories of white blood cells: myeloid and lymphoid.
Myeloid cells
There are three broad groups of myeloid cells: granulocytes, monocytes and mast cells. They are all produced in a process called myelopoiesis in the bone marrow.
Granulocytes make up the largest proportion of white blood cells in the circulation. They are called as such due to the presence of granules in their cytoplasm. Granulocytes include:
- Neutrophils: the first type of white blood cell to arrive at a site of inflammation through the mechanism of chemotaxis. They are involved in the phagocytosis of extracellular pathogens. They contain granules that enable them to kill these pathogens with proteolytic enzymes and myeloperoxidase. On blood films they have multi-lobulated nuclei.
- Eosinophil: present at a much lower level than neutrophils. They are elevated in hypersensitivity reactions and in response to parasitic infection. On a blood film they have pink-staining granules in the cytoplasm and have a kidney shaped nucleus. The granules contain histamine, leukotrienes and other molecules that help with phagocytosis.
- Basophils: represent the smallest proportion of white cells. They are elevated in inflammatory and allergic reactions. The granules contain histamine and heparin which induce vasodilation and increased vessel permeability. They are more blue on staining and tend to obscure the nucleus.
Mast cells are found in tissue (e.g. interstitial connective tissue and submucosa) rather than blood and have similarities to basophils in that they contain basophilic granules and receptors for IgE antibodies. They are involved in mediating the type I hypersensitivity reaction.
Monocytes do not remain in the circulation for very long and differentiate into macrophages when they migrate into the tissues. If they are found in the peripheral blood, they are large leukocytes with a horseshoe shaped nucleus. Macrophages differentiate further upon reaching their target tissues, such as Kupffer cells in the liver and microglial cells in the nervous system. Macrophages’ main role is phagocytosing pathogens, debris and dead cells and presenting antigens from the debris to lymphocytes, which will commence the adaptive response.

Lymphoid cells
Lymphoid cells include T cells, B cells and natural killer (NK) cells. T cells mature in the thymus (hence ‘T’) and B cells mature in the bone marrow. These cell types undergo several mechanisms during development, including somatic recombination and tolerance, which allow them to recognise multiple different epitopes whilst at the same time ignoring self-antigens. NK cells are part of the innate immune system and therefore do not require these processes.
T cells are involved with the cell-mediated immune response. There are many subtypes but the two main ones are CD4 and CD8. CD8 cells are cytotoxic and kill cells directly, whereas CD4 T cells help activate B cells which recognise the foreign antigen and, therefore, produce antibodies.
B cells reside in the lymphoid tissue after they have matured. After they are activated by T cells, they will become plasma cells and memory B cells. Plasma cells secrete antibodies to fight the infection. Memory B cells remain in the body for years after infection and are primed to respond to the pathogen if it enters the body again.
Lymphocytes have a large round nucleus with little cytoplasm around the age with no granules. It is not possible to discern which are T or B cells on a blood film.
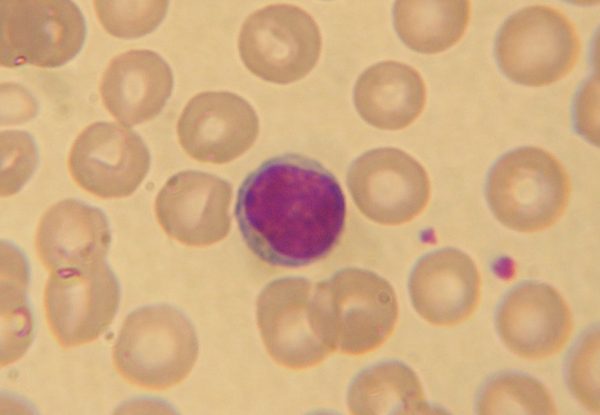
Source: No machine-readable author provided. NicolasGrandjean assumed (based on copyright claims)., CC BY-SA 3.0, via Wikimedia Commons
Platelets
Platelets, also known as thrombocytes, are fragments of megakaryocytes which are involved in clotting of the blood. As they do not contain a nucleus, they are not considered true cells. They exist in the circulation for 5-7 days. Their main function is haemostasis and thrombosis. Platelets contain granules which initiate the clotting cascade, particularly von Willebrand factor, fibrinogen, and platelet factor 4. They have several surface proteins which allow them to aggregate and bind to blood vessel walls.
Sometimes the formation of a thrombus can be pathological, classically in coronary heart disease. They are targeted with antiplatelet drugs such as clopidogrel, ticagrelor and prasugrel for treatment of cardiovascular disease. These drugs target receptors on platelets and decrease their aggregation and haemostasis. As a result, they prevent arterial thrombosis.

Source: Keith Chambers, CC BY-SA 3.0, via Wikimedia Commons
Plasma
Plasma contains a variety of different substances, such as albumin, immunoglobulins, coagulants and electrolytes. It is the liquid component of blood. Without it, blood could not perform many of its functions, such as coagulation, immune function and nutrition. Albumin is the main protein in the plasma and has a primary role in maintaining oncotic pressure and transporting substances.
Plasma can be separated from the cellular components of blood and stored as fresh frozen plasma, which is given in major haemorrhage.
Haematopoiesis
Haematopoiesis is a process that occurs in the bone marrow where new blood cells form from pluripotent haematopoietic stem cells. Throughout life, there is rapid turnover of blood cells , so haematopoiesis plays a crucial role in maintaining oxygen delivery and maintenance of the immune system.
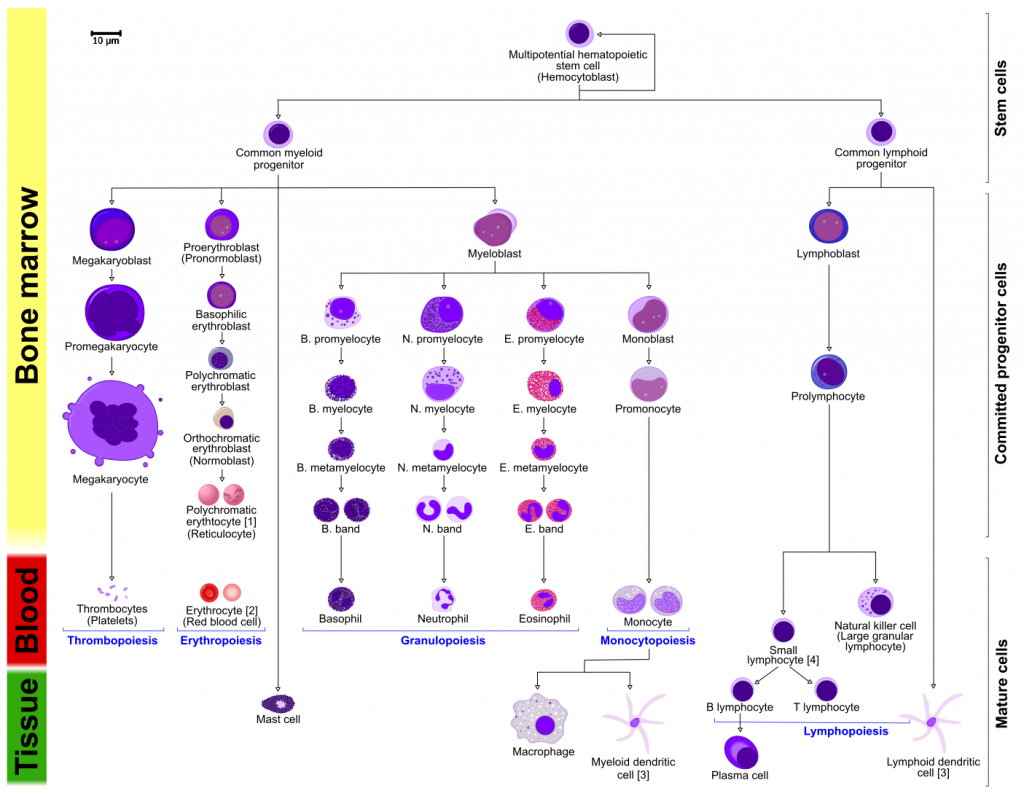
Source: !Original: A. RadVector: RexxS, Mikael Häggström and birdy and Mikael Häggström, M.D. - Author info - Reusing images, CC BY-SA 3.0, via Wikimedia Commons
There are two broad progenitor lineages: myeloid and lymphoid. The progenitor cells have the suffix ‘blast’. Understanding these lineages is essential for understanding malignancies as you will understand how a specific disease may manifest by knowing the cell origin.
Myeloid splits into three different routes:
- Platelets
- Red blood cells
- White myeloid cells, including granulocytes and agranulocytes
Lymphoid broadly splits into lymphocytes and natural killer cells.
Blood cell maturation
The production of blood cells is continuous throughout one’s life, and therefore varying the rate of production makes the system much more adaptable to environmental pressures. One way the haematological system is regulated is with haematopoietic growth factors.
Erythropoiesis is initiated by erythropoietin from the kidneys. This growth factor promotes differentiation of stem cells to the erythroid cell line. The stage before a mature erythrocyte is a reticulocyte and these may be present in the circulation in certain pathological conditions.
Thrombopoiesis, the production of platelets, is initiated by thrombopoietin, which triggers development of megakaryoblasts.
Granulopoeisis, which leads to the production of granulocytes, begins with Granulocyte macrophage colony stimulating factor (GM-CSF); this promotes differentiation of the common myeloid precursor to the myeloblast.
Monopoeisis is promoted by monocyte colony stimulating factor (M-CSF).
Lymphocytes all originate in the bone marrow but have secondary sites of maturation which reflect their different functions in the adaptive immune system.
Bone marrow
Bone marrow fills the cavity within bone and has a very different consistency, being a more jelly-like substance. There are two key types of bone marrow:
- Red marrow: highly vascular and haematopoietically active. Red marrow is present in all skeletal structures until the 5th year of life. From then onwards it becomes restricted in the central flat bones and proximal ends of long bones. Supporting cells are required in the red marrow, such as reticular cells and macrophages. Reticular cells produce reticulin that support the haematopoietic cells in the marrow. Macrophages clear cellular debris to enable efficient haematopoiesis.
- Yellow marrow: less haematopoietically centred and contains more adipocytes. Yellow marrow takes up a bigger proportion of the bone marrow with age. Reticular cells take up lipid and this begins the conversion to yellow marrow. The number of adipocytes increase as well as reduction in haematopoietic centres.
In many haematological conditions the bone marrow may be assessed to get a fuller picture of what is going on with the patient. It allows us to assess if the bone marrow is healthy or dysfunctional, and for what reason that may be, be it infiltrative disease, depletion of progenitor cells and/or nutrient deficiencies.
References
- https://www.britannica.com/science/blood-biochemistry
- https://www.amboss.com/us/knowledge/Basics_of_hematology
- https://www.kenhub.com/en/library/anatomy/histology-of-bone-marrow
- https://www.kenhub.com/en/library/anatomy/histology-of-hematopoiesis
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5709181/
- https://www.ncbi.nlm.nih.gov/books/NBK531504/
- https://www.kenhub.com/en/library/anatomy/cells-of-the-blood








Superb notes
Notes are well organised and easy to comprehend