Introduction
The renal system plays an essential homeostatic function through urine production and alterations in:
- Water handling
- Solute regulation, including the crucial control of potassium levels
- Acid-base regulation
Urine production
Glomerular filtration
Renal blood flow accounts for 25% of cardiac output. This provides a hydrostatic pressure gradient to drive ultrafiltration at the glomerulus.
Glomerular filtration rate (GFR) is the flow rate of filtered fluid through the kidney. Filtration is dictated by the following Starling force’s law equation:
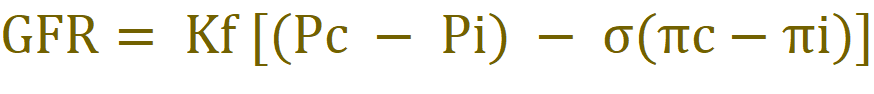
Pc: Capillary hydrostatic pressure
Pi: Bowman’s capsule hydrostatic pressure
σ: Reflection coefficient
πc: Capillary oncotic pressure
πi: Bowman’s capsule hydrostatic pressure
Filtration and reflection coefficients are generally fixed values, so the degree of filtration is dictated by hydrostatic and oncotic pressures. The kidney primarily regulates filtration through alterations in renal blood flow.
Regulation of GFR
GFR is maintained at ~ 125 ml/min by changing capillary hydrostatic pressure and renal blood flow. This can be achieved through regulation of the afferent and efferent glomerular arterioles:
- Afferent arteriole constriction → ↓ Pc → ↓ GFR + ↓Renal blood flow
- Efferent arteriole constriction → ↑ Pc → ↑ GFR + ↓ Renal blood flow
The kidneys maintain a relatively constant renal blood flow while altering glomerular filtration rate. This can be achieved by altering afferent and efferent arteriolar diameters as they have antagonistic effects on glomerular filtration but synergistic effects on renal blood flow.
Mechanisms used to monitor and produce feedback regulation include:
- Myogenic mechanism (arteriole contraction on stretching)
- Tubulo-glomerular feedback: Macula densa releases ATP in response to increased NaCI, producing afferent arteriole constriction (thereby reducing filtration)
- Angiotensin II: Produces efferent arteriole constriction
- Sympathetic nerve stimulation: Produces afferent arteriole constriction
Solute reabsorption
The proximal convoluted absorbs most solutes as well as all glucose & amino acids. Therefore, the distal parts are responsible for the regulation of solute reabsorption. A broad summary of the function of different parts of the nephron is:
- Proximal convoluted tubule: fluid reabsorption
- Loop of Henle: regulates urine concentration
- DCT: pH and potassium regulation
- Collecting ducts: Can concentrate urine
Transporters
The nephron has wide range of different transporters. Clinically significant transporters include:
- Proximal convoluted tubule: Na+ / H+ exchanger
- Thick ascending loop of Henle: Na-K-2Cl cotransporter
- Distal convoluted tubule: Na-Cl cotransporter
- Collecting duct: ENaC channel
Water handling
Plasma osmolality is kept at 280-300 mOsm/kg by regulating urine concentration. The kidneys can produce urine concentrations ranging from 30-1200 mOsm/kg.
Urinary concentration is mainly controlled by the antidiuretic hormone (ADH) released by the posterior pituitary. Regulation of urinary concentration occurs after the loop of Henle; at this point, tubular fluid is hypo-osmotic and can subsequently:
- Produce dilute urine: Without ADH, there is low water permeability in the distal convoluted tubule and collecting tubes. Thus, solutes continue to be absorbed, making urine hypo-osmolar
- Produce concentrated urine: ADH increases water permeability, allowing water to be reabsorbed into the interstitium.
Potassium regulation
Factors affecting potassium regulation
Mechanisms that produce high potassium via cellular shifts include:
- Dehydration → Cell shrinkage → ↑ Intracellular potassium concentration → ↑Shifting of potassium to extracellular compartments
- Cell lysis, leading to release of intracellular potassium
- Acidosis, which leads to increased release of potassium from intracellular to extracellular compartments via cellular K+/H+ exchanger as a homeostatic mechanism to counteract acidosis
On the other hand, mechanisms producing low potassium include:
- Hyperhydration
- Alkalosis
- Insulin, which stimulates cellular Na/K exchange
- Adrenaline, which stimulates cellular Na/K exchange
Note that insulin and adrenaline have the above actions as part of a physiological feedforward response to hyperkalaemia as a result of eating and exercise, respectively.
Cellular effects of potassium change
High potassium levels increase the excitability of cells by shifting resting membrane voltage closer to the threshold potential and, therefore, increases the possibility of depolarisation.
Conversely, low potassium levels hyperpolarise the cell, thereby reducing cellular excitability.
Regulation of potassium via kidneys
Potassium is mainly regulated in the distal convoluted tubule and collecting ducts via the following mechanisms:
- Potassium plasma concentration alters the activity of the Na/K exchanger
- Aldosterone: stimulates ENaC production, Ca-activated K channels and Na/K exchanger, producing an overall increase in potassium excretion

Acid-base regulation
Short-term control
Physiological buffers include haemoglobin, plasma proteins and phosphates that act as static, short-term buffers. In addition, a buffering system based on bicarbonate is also employed and has the benefit of being dynamic and more robust.

However, this bicarbonate buffering system is not sustainable without the kidneys as it relies on regeneration of bicarbonate.
Long-term control
Usually, kidneys are not producers of bicarbonate. This achieved by:
- HCO3 reabsorption by H+ secretion into tubules.
- Production of HCO3 from glutamine via ammoniagenesis








0 Comments