What is anaemia?
Anaemia is the deficiency of haemoglobin in the blood. The thresholds are different for men and women.
| Sex | Normal range (g/l) |
| Man | 135-175 |
| Woman | 115-155 |
Anaemia itself is not a diagnosis and instead is usually a result of another condition.
Presentation of anaemia
Anaemia affects a large demographic of people due to the wide variety of causes. Typically, in mild forms of anaemia patients are unlikely to have any symptoms directly associated with the low haemoglobin. It is only when levels drop significantly, generally below 80g/l, do the following symptoms occur:
- Tiredness
- Breathlessness on exertion
- Dizziness
- Chest pain
- Palpitations
- Appearing paler than usual
If the anaemia is of more rapid onset then symptoms are more severe than slower onset, as the body has less of a chance to adapt.
Classification of anaemia
Anaemia can be classified by the mean corpuscular volume (MCV), which is included in a full blood count blood test. The MCV is the average size and volume of a red blood cell. The normal range varies between hospitals as it depends on the equipment in their respective laboratories. Nonetheless, here is a general range:
| Description | Mean corpuscular volume (fL) |
|---|---|
| Microcytic | <80 |
| Normocytic | 80-100 |
| Macrocytic | >100 |
Classifying according to MCV is a helpful way of organising anaemia as it can subdivide the many different causes of anaemia.
Microcytic anaemia
Microcytic anaemia is anaemia with blood cells smaller than normal. The four top causes can be remembered with a helpful mnemonic: ‘Find Those Small Cells’.
Fe (Iron deficiency)
Thalassaemia
Sideroblastic anaemia
Chronic disease
Iron deficiency anaemia
Iron is an essential component of haemoglobin in red blood cells. Iron deficiency anaemia (IDA) is the commonest cause of anaemia in the world. It occurs when the body does not have enough iron stores to produce sufficient erythrocytes. Women are more susceptible to this than men due to loss of blood in their menstrual cycle. The causes of IDA can be split into four categories – reduced intake, increased utilisation, blood loss and reduced absorption.
| Category | Causes |
|---|---|
| Reduced intake | Diet (vegetarians, vegans, malnutrition) |
| Increased utilisation | Pregnancy, growth spurts in children |
| Blood loss | menstruation, peptic ulcers, cancer, parasitic infection |
| Malabsorption | Coeliac disease, inflammatory bowel disease, H pylori colonisation, gastrectomy, medications |
Risk factors can be drawn from the causes:
- Menorrhagia (heavy periods)
- Vegetarian/vegan diet
- Hookworm infection
- Coeliac disease
- Use of Non-steroidal anti-inflammatory
- Pregnancy
On a blood film, the red blood cells look smaller (microcytic) and paler (hypochromic) as they have less haemoglobin. There is also a variation in red blood cell size (anisocytosis) and shape (poikilocytosis), which is typical for iron deficiency anaemia. Also seen in iron deficiency anaemia but not depicted below are pencil poikilocytes which are elongated and flattened red blood cells.

Presentation
Patients with anaemia are generally asymptomatic until the anaemia becomes particularly severe. Along with the symptoms listed previously, patients with IDA may specifically develop:
- Atrophic glossitis
- Angular Stomatitis
- Koilonychia
- Pica – craving non-food substances such as ice or dirt
- Restless leg syndrome
- Rarely, tachycardia and signs of heart failure
More commonly, patients will present with symptoms of the underlying cause of IDA.
| Symptom | Condition |
|---|---|
| Dysphagia | Oesophageal malignancy, Plummer-Vinson syndrome |
| Dyspepsia | Gastric cancer, peptic ulcer disease |
| Abdominal pain | Coeliac disease, intra-abdominal malignancy, inflammatory bowel disease |
| Change in bowel habit | Bowel cancer, coeliac disease, inflammatory bowel disease |
| Rectal bleeding | Anal fissure, rectal cancer, haemorrhoids, inflammatory bowel disease |
| Weight loss | Inflammatory bowel disease, coeliac disease, bowel cancer |
Investigations
When a patient is found to have anaemia, it is important to do some baseline investigations to find the cause of it.
These include:
- Full blood count
- Haemoglobin
- MCV
- Red cell distribution width – variation in size of red blood cells which is increased in IDA
- Blood film
- Iron studies
- B12 and folate – more about this later
Other investigations that could be considered depend on the demographic of the patient. These will be unnecessary in patients where there is an obvious cause of iron deficiency, such as a pregnant woman with no other co-morbidities or a young woman with menorrhagia.
When this is not the case, the following investigations may be carried out:
- Coeliac serology: tissue transglutaminase antibody
- Urine dipstick: to screen for haematuria
- Stool examination: detecting parasites
- Endoscopy/colonoscopy to exclude chronic blood loss from gastrointestinal malignancy in patients who have at least one of:
- Over 60 years old
- Premenopausal women with bowel symptoms
- Family history of GI cancer
- Persistent anaemia despite treatment
A word on iron studies
Iron studies is a blood test that looks at the following four measurements:
- Transferrin saturation
- Total iron binding capacity (TIBC)
- Serum iron
- Ferritin
Transferrin is the iron transport protein in the plasma. It is increased in iron deficiency to maximise the use of the available iron in the body. Transferrin saturation refers to the amount of transferrin bound to iron. Unsurprisingly, this falls in iron deficiency.
TIBC refers to the availability of iron binding sites on transferrin. As there is less iron in the body but more transferrin, this value is increased in IDA.
Serum iron is the measure of ferric ions (Fe3+) in the blood. It varies a lot and is not a reliable indicator of IDA.
Ferritin is the form iron takes when it is in cells. Typically, it will be low in IDA. However, you may remember that it is one of the acute phase proteins, hence it is increased in general inflammation. Therefore, it may be falsely high in patients with an infection or malignancy.
Interpretation can be difficult, so it will take some practice. Here is a table summarising:
| Iron deficiency | Anaemia of chronic disease | Acute phase reaction | Iron overload | |
|---|---|---|---|---|
| Serum iron | ↓ | ↓ | ↓ | ↑ |
| Transferrin/TIBC | ↑ | ↓ | ↓ | ↑ |
| Transferrin saturation | ↓ | ↓ | ↓ | ↑ |
| Ferritin | ↓ | ↔ | ↓ | ↑ |
Management
If the iron deficiency anaemia is a result of diet, then iron supplementation should be prescribed. Typically, this will be oral ferrous sulphate and treatment should be continued for at least three months. Unfortunately, iron tablets have their own side effects, such as nausea and constipation, which the patient should be counselled about. An FBC should be done up to 4 weeks after to monitor the effect. If the haemoglobin has not improved, then alternative causes of the anaemia will need to be explored.
Intravenous iron is rarely used and will only be considered if there is a genuine intolerance to oral iron or a more rapid response is required.
Blood transfusions are almost never required. They should be reserved for cases of severe and/or symptomatic anaemia as receiving a blood transfusion comes with its own morbidities.
If an underlying cause is found or suspected, then part of the management is referral to the relevant specialty.
Anaemia of Chronic disease
Anaemia of chronic disease is another type of microcytic anaemia which occurs in chronic inflammatory states. In this condition, the body releases cytokines and acute phase proteins. One of these is called hepcidin, which works to sequester iron into cells. This is an evolutionary adaptation as bacteria utilise iron to replicate; if bacteria cannot access iron, it is harder for them to build large enough colonies to cause infection. If this goes on for a long time, iron sequestration can then affect the production of erythrocytes and this leads to anaemia.
Causes of anaemia of chronic disease include:
- Autoimmune disorders, such as rheumatoid arthritis
- Chronic infections
- Chronic diseases, such as chronic kidney disease and heart failure
- Malignancy
Presentation
Patients with anaemia of chronic disease will unlikely present with symptoms of anaemia. Anaemia is more likely to be found incidentally, especially as the symptoms for anaemia will overlap with the symptoms of the disease they’re suffering from.
Investigations
These will be very similar to those above. See table 5 for interpretation of iron studies in anaemia of chronic disease.
Anaemia of chronic disease is a diagnosis of exclusion, hence other causes would be looked for first before concluding this as the diagnosis.
Management
Treatment of the underlying condition is prioritised first and the anaemia may correct itself with adequate control. The body experiences a functional deficiency of iron rather than absolute deficiency but iron supplementation may still be considered. It is also important to note that multiple causes may be causing a patient’s anaemia, so they may also be concurrently iron deficient.
Sideroblastic anaemia
This is a group of disorders characterised by dysfunctional haemoglobin synthesis in the mitochondria. This leads to the formation of ring sideroblasts in the cytoplasm and microcytic cells secondary to the disease pathogenesis. Causes include genetic enzyme deficiency, and environmental causes: vitamin B6 deficiency, lead poisoning and chronic alcoholism.
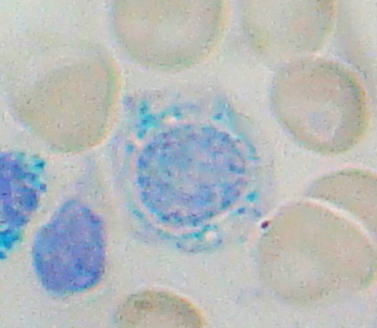
Management is generally supportive, with consideration of transfusion for severe anaemia. Iron chelation may be considered to reduce formation of sideroblasts.
Thalassaemias
Thalassaemias are a group of autosomal recessive inherited conditions characterised by an absence of one of the globin genes involved in haemoglobin formation. As a result, the haemoglobin structure is affected, which is intimately linked with its function. The two main types are alpha and beta thalassaemia depending on which globin genes are lost.
Pathophysiology
When one of the globin genes is missing, there is accumulation of the excess chain type in red blood cells which predispose them to oxidative damage and therefore cell death. This leads to a slight haemolytic anaemia which is seen in these patients. In addition, there is reduced haemoglobin synthesis in general which leads to the microcytic nature of the anaemia.
Presentation
Symptom severity is correlated with the severity of the thalassaemia. Therefore, for more severe forms, they may be symptomatic within a few months after birth while less severe forms can present in young adulthood. Generally, patients will present with symptoms of anaemia (fatigue and pale). Due to the excessive haemolysis secondary to oxidative damage, jaundice may also be apparent. In beta thalassaemia major the bone marrow expands to compensate for the ineffective erythropoiesis leading to jaw and forehead expansion – ‘thalassaemia facies’. These individuals may also be prone to more infections. On examination they may have hepatomegaly or splenomegaly.
Investigations
A FBC would show a microcytic anaemia. Iron studies would show an elevated iron level with a raised ferritin, although that is non-specific.
The most helpful investigation is haemoglobin electrophoresis which usually reveals the diagnosis. It shows the percentage of different types of haemoglobin, which are formed by different globin combinations, and excessive amount of a particular type confirms the thalassaemia.
Imaging that may be considered include a plain skull X-ray. In beta thalassaemia major it will show a classical ‘hair on end’ appearance, with overgrown maxilla and separation of the orbit.
Bone marrow biopsy may be considered in other conditions that may mimic thalassaemia major’s presentation.
Management
Asymptomatic carriers have little management required apart from monitoring them for iron deficiency. Transfusions may be required for more severe disease to keep haemoglobin above a certain level. These individuals may also be counselled prior to having children to assess the risk of passing down the genes.
Normocytic anaemia
Normocytic anaemia is low haemoglobin when the MCV is within the normal range. The cause of this can be broken down into increased destruction or decreased production. Increased destruction suggests good bone marrow function, whereas reduced production suggests poor bone marrow function.
| Increased destruction | Decreased | |
|---|---|---|
| Hereditary | Hereditary spherocytosis Sickle cell anaemia Glucose-6-phosphate dehydrogenase deficiency Pyruvate kinase deficiency | Chronic renal failure |
| Acquired | Autoimmune haemolytic anaemia Paroxysmal nocturnal haemoglobinuria Micro/macroangiopathic haemolytic anaemia Acute blood loss | Anaemia of chronic disease Aplastic anaemia Myelophthisic process |
Table 6. Types of normocytic anaemia
Decreased production
Decreased haemoglobin production suggests the bone marrow is affected.
In chronic renal failure, the cause centres on erythropoietin secretion. Normally the kidney produces erythropoietin in response to blood oxygen concentration. When hypoxia occurs, more of the interstitial cells in the kidney produce erythropoietin which acts on the bone marrow to stimulate erythropoiesis. However, when the kidneys are losing their function the erythropoietin secreting cells are fewer and, therefore, less is secreted overall. As a result, fewer red blood cells are produced and anaemia occurs.
A myelophthisic process is a process which involves infiltration of the bone marrow. This can include a variety of conditions such as haematological cancers, lymphomas and myelofibrosis. Myelophthisis reduces the area in the bone marrow where erythropoiesis can occur, hence fewer erythrocytes are produced.
Aplastic anaemia is a rare group of conditions wherein haematopoietic stem cells numbers are reduced. Other cell lines may also be affected aside from red blood cells. It can be idiopathic, autoimmune or drug-related.
Anaemia of chronic disease can also produce normocytic anaemia (or, in some cases as mentioned previously, microcytic anaemia).
Increased destruction
Excessive breakdown of red blood cells that exceeds the rate of their production can produce normocytic anaemia.
Haemolysis of red blood cells is a physiological process and is always extravascular. This is necessary to recycle components of the red blood cells to be reused in new erythrocytes. These components are:
- Haem → recycled into iron
- Globin → recycled into amino acids
- Protoporphyrin → recycled into unconjugated bilirubin which has its own subsequent pathway
In pathological haemolysis, the breakdown of red blood cells exceeds the rate of their production. Also, haemolytic anaemias can be both intravascular or extravascular.
There are some clinical features specific to haemolytic anaemias, including:
- Jaundice
- Splenomegaly
- Gallstones
- Aplastic crisis after parvovirus infection
Inherited causes lead to excessive haemolysis because their abnormal structure of red blood cells means they are more susceptible to breakdown. They can split up into the site of the inherited defect:
- Membrane abnormalities (e.g. hereditary spherocytosis)
- Metabolic deficiencies (e.g. G6PD deficiency)
- Haemoglobin abnormalities (e.g. alpha-thalassaemia, beta-thalassaemia, sickle cell disease)
Acute blood loss can also be a cause of normocytic anaemia.
Investigations
Baseline blood tests that should be done include full blood count (FBC), blood film, liver function tests (LFTs), lactate dehydrogenase (LDH), haematinics and iron studies. Bilirubin and LDH are elevated in a haemolysis picture due to increased cell breakdown. Reticulocytosis may be seen, which is the increase of the red blood cell progenitor, the reticulocyte, in the circulation. This occurs as the bone marrow attempts to compensate for the excessive RBC breakdown.
Blood films may be very informative in cases of congenital red blood cell defects.
Subsequent tests may include a bone marrow biopsy, which would clinch the diagnosis in a myelophthisic process. Other tests that could be done are specific to the cause, such as serology to look for autoantibodies, enzyme levels for congenital defects, and membrane tests.
Management
Management should be focused on the underlying condition. Once that is corrected then the anaemia will usually improve. If the cause is acute blood loss, then a blood transfusion may be required.
Macrocytic anaemia
Macrocytic anaemia is anaemia with an MCV >100fl. Another helpful mnemonic for remembering the causes of this is FAT RBC.
Foetus (pregnancy)
Alcohol excess
Thyroid (hypo)
Reticulocytosis
B12 and folate deficiency
Chronic liver disease
The causes can be subdivided further into megaloblastic and normoblastic, which refers to the maturation state and appearance of the blood film. Megaloblastic anaemia is typically caused by vitamin B12 and folate deficiencies.
Megaloblastic anaemia: B12 and folate deficiency
In megaloblastic anaemia, erythroblasts (progenitors of red blood cells) show characteristic delayed maturation of nucleus relative to cytoplasm caused by defective DNA synthesis. On a blood film you may see hypersegmented neutrophils (neutrophils with 5 or more nuclei segments).
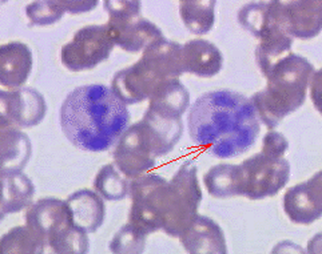
The most common causes are B12 and folate deficiencies; other causes include cytotoxic drugs, which inhibit the DNA synthesis pathways, and inborn errors of metabolism.
Pathophysiology
Folic acid and vitamin B12 or cobalamin are required in DNA synthesis. Folate is specifically required for producing thymidine, one of the bases. Vitamin B12 is required as a co-factor in the Krebs cycle and in the regeneration of tetrahydrofolate. Deficiency leads to ineffective erythropoiesis in the bone marrow and enlarged red blood cells. These deficiencies also affect other cell lines such as neutrophils, producing the hypersegmented nuclei.
The causes of each respective deficiency are listed on the table below.
| Folate deficiency | Vitamin B12 deficiency |
|---|---|
| Inadequate dietary intake Malabsorption e.g. coeliac disease, Crohn’s disease, phenytoin, oral contraceptive pill, alcohol Increased requirement e.g. pregnancy Increased loss e.g. in urine Drug inhibition: methotrexate and trimethoprim (inhibit dihydrofolate reductase) | Inadequate intake: malnutrition and vegan diet Inadequate intrinsic factor - Pernicious anaemia - Gastrectomy Malabsorption Diversion of dietary vitamin B12 - Abnormal intestinal bacterial flora Increased requirement: pregnancy and lactation |
Vitamin B12 deficiency
B12 is found in meat and dairy products. Therefore, deficiency may be found in long-term vegans. Vitamin B12 stores last for years before deficiency develops.
Normal absorption of vitamin B12 requires a glycoprotein called intrinsic factor (IF). Intrinsic factor is normally secreted by parietal cells. The B12-IF complex binds to receptors within the terminal ileum where it is absorbed. Pernicious anaemia occurs when this process is disrupted due to autoantibodies destroying the parietal cells or intrinsic factor itself.
When vitamin B12 deficiency is long standing or severe, neuropsychiatric complications can develop. These include:
| Neurological features | Psychiatric features |
|---|---|
| Peripheral neuropathy Subacute degeneration of the cord Focal demyelination | Depression Personality change Memory loss |
Folate deficiency
Folate is found in green leafy vegetables, pulses and cereals. Unlike vitamin B12, folate stores only last a few months and is lost more quickly. Patient with malnourishment are susceptible to developing folate deficiency, as well as pregnant women.
Normal absorption of folate occurs in the proximal part of the small intestines. Conditions that affect this area of the bowel, such as coeliac disease, can lead to deficiency.
Presentation
Aside from the normal symptoms of anaemia, patients may present with the neuropsychiatric symptoms as seen before. They may also be mildly jaundiced due to increased cell turnover. On blood tests they may have low white cells or platelets as folate and vitamin B12 deficiencies can affect other cell lines.
Investigations
Blood tests that would be done would be FBC, LFTs LDH, blood film, haematinics, vitamin B12 and folate levels. Bilirubin and LDH become elevated in increased red cell breakdown, which is seen in other disorders, but in the case of megaloblastic anaemia indicates excessive destruction in the bone marrow. Red cell folate is a more accurate measure of levels than serum folate. Haematinics looks specifically at vitamin B12, folate, ferritin and intrinsic factor, deficiencies of which are common causes of anaemia.
If B12 levels were low, subsequent tests would include serology to look for autoantibodies seen in pernicious anaemia and gastroscopy.
Treatment
The first-line treatment is to replace the deficient vitamins. It is crucial to remember vitamin B12 must be replaced before folate; folate supplementation alone can drain existing vitamin B12 stores and lead to neurological complications (subacute combined degeneration of the spinal cord).
Vitamin B12 is typically replaced with intramuscular injection of 1mg, 3 times a week for two weeks. This is followed by maintenance of 1mg every 3 months.
Folate is replaced with 5mg oral tablets daily for 4 months.
Non-Megaloblastic causes
The remainder of the causes of macrocytic anaemia in the mnemonic are non-megaloblastic, meaning DNA synthesis and erythropoiesis is not affected.
Alcohol has a toxic effect on erythroblasts and sometimes a large MCV can be a good indicator a patient is still drinking alcohol.
Liver disease, of different causes, can cause macrocytosis due to its effect on lipid production required for membrane synthesis.
Hypothyroidism can also cause macrocytic anaemia.
Reticulocytosis is when the progenitor of red blood cells, reticulocytes, are increased in the circulation. These are larger than mature blood cells, hence can shift the average MCV to be larger than normal. It is a clinical feature of several blood disorders, such as haemolytic anaemia.
Another cause not listed in the mnemonic is certain medications, such as cyclophosphamide and azathioprine, which inhibit metabolic pathways required for erythropoiesis.
Treatment will ultimately be focused the underlying condition, which in turn should lead the macrocytic anaemia to correct itself.
References
- https://geekymedics.com/iron-deficiency-anaemia/
- https://www.ncbi.nlm.nih.gov/books/NBK545275/
- https://www.bmj.com/content/357/bmj.j2513.full
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5588399/
- https://app.pulsenotes.com/medicine/haematology/notes/macrocytic-anaemia
- https://patient.info/doctor/thalassaemia-pro








0 Comments